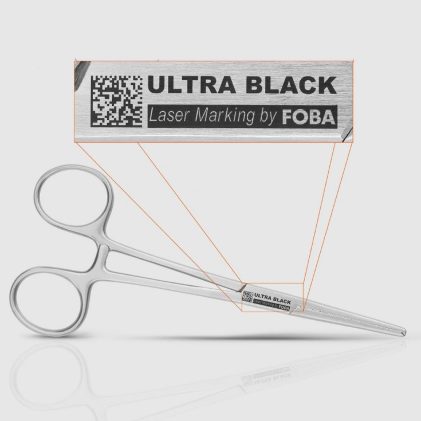
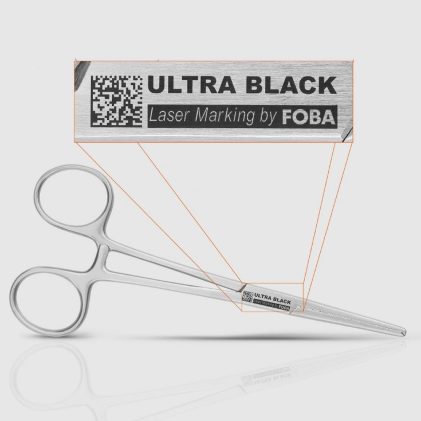
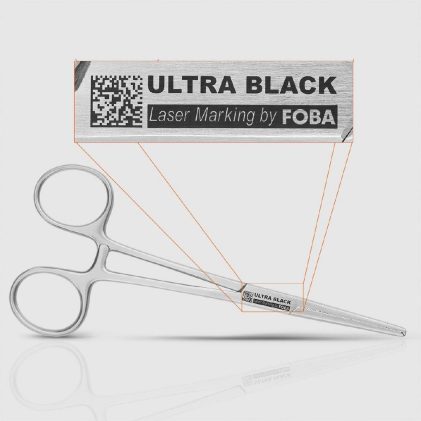
With UDI requirements under FDA regulations and the EU MDR, direct part marking has become mandatory for many medical devices, including surgical instruments and implants. In practice, these requirements collide with highly reflective metals, extremely limited marking areas, and demanding conditions across the entire device lifecycle. Markings must remain permanently high‑contrast and reliably legible without […]
Read More